Rebuilding a Brittle Market: Regulatory Reform and the Return of Innovation in Osteoporosis
By Luma Group - 5 minutes
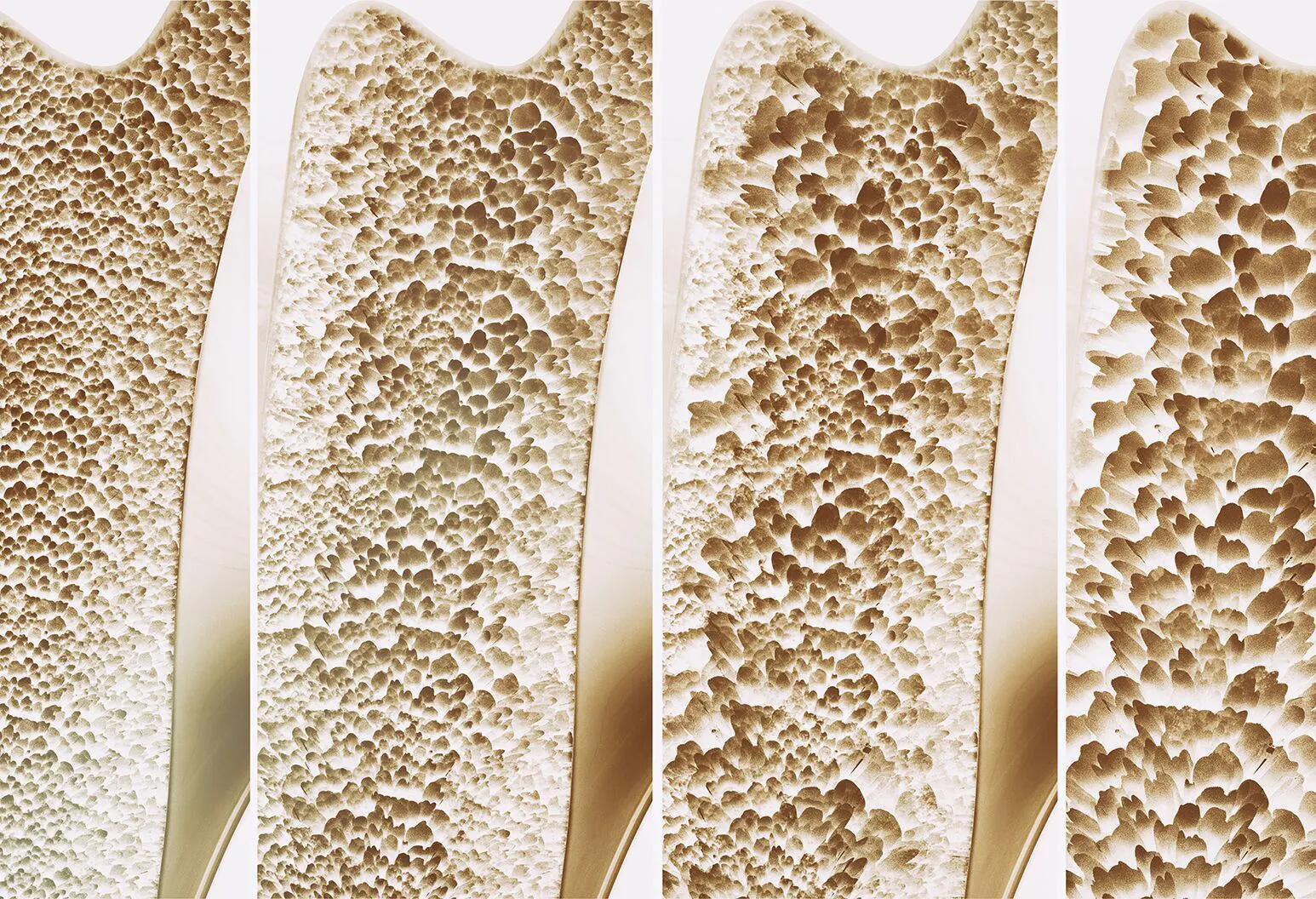
Osteoporosis, a condition where bones become weak and brittle due to loss of bone density, is often seen as a natural part of aging, but it is more accurately a chronic disease caused by disrupted bone remodeling.
10 million Americans over age 50 have osteoporosis, and another 43 million have low bone mass.[1] Despite this, many patients remain undiagnosed and untreated. While existing therapies can reduce fracture risk, they do not provide a safe, effective long-term solution for a disease that develops over decades.
Progress in the field has been slowed not only by biology but also by demanding regulatory requirements that de-incentivized innovation. Historically, the FDA required large, lengthy, and expensive phase 3 trials. These trials often involved more than 7,000 patients over 3–5 years to show reductions in fracture risk, even when improvements in bone mineral density (BMD) were observed.
In December 2025, the FDA updated its approach, accepting total hip BMD, measured by dual-energy X-ray absorptiometry (DXA), as a validated surrogate endpoint for postmenopausal osteoporosis trials. [2],[3] While this does not solve the underlying biology, it significantly reduces the time, cost, and complexity of clinical development, enabling clinical proof-of-concept in a 24-month trial and making osteoporosis a more attractive area for innovation and investment.
April 2026
Scientific Background, Current Standard of Care, and Treatment Gaps
Osteoporosis is not a passive “bone thinning” process, but an active dynamic remodeling disorder in living bone tissue. In healthy adults, bone remodeling is continuous: osteoclasts cells remove old or microdamaged bone, osteoblasts cells replace it with new bone, and the net balance preserves skeletal mass and mechanical integrity. Osteoporosis develops when that balance is persistently shifted toward bone resorption or away from adequate bone formation.
The disease disproportionately affects women, as 1 in 3 over the age of 50 experience fractures, compared to just 1 in 5 men. The sudden drop in estrogen during menopause is the key culprit for this gender disparity, as estrogen is a key regulator that orchestrates the balance between osteoblasts and osteoclasts. In total, women lose up to 90% of their estrogen in a span of 4-5 years during perimenopause. This sudden estrogen decline pushes the RANKL-OPG signaling pathway axis to favor osteoclastogenesis, amplifying pro-inflammatory signaling in the bone marrow microenvironment and contributing to impaired osteoblast function through osteocyte and Wnt-pathway biology. Clinically, this often unfolds in two phases: an accelerated early phase of bone loss over roughly the first 5 to 10 years after menopause, followed by a slower but indefinite phase that progressively affects both trabecular and cortical bone.[4],[5]
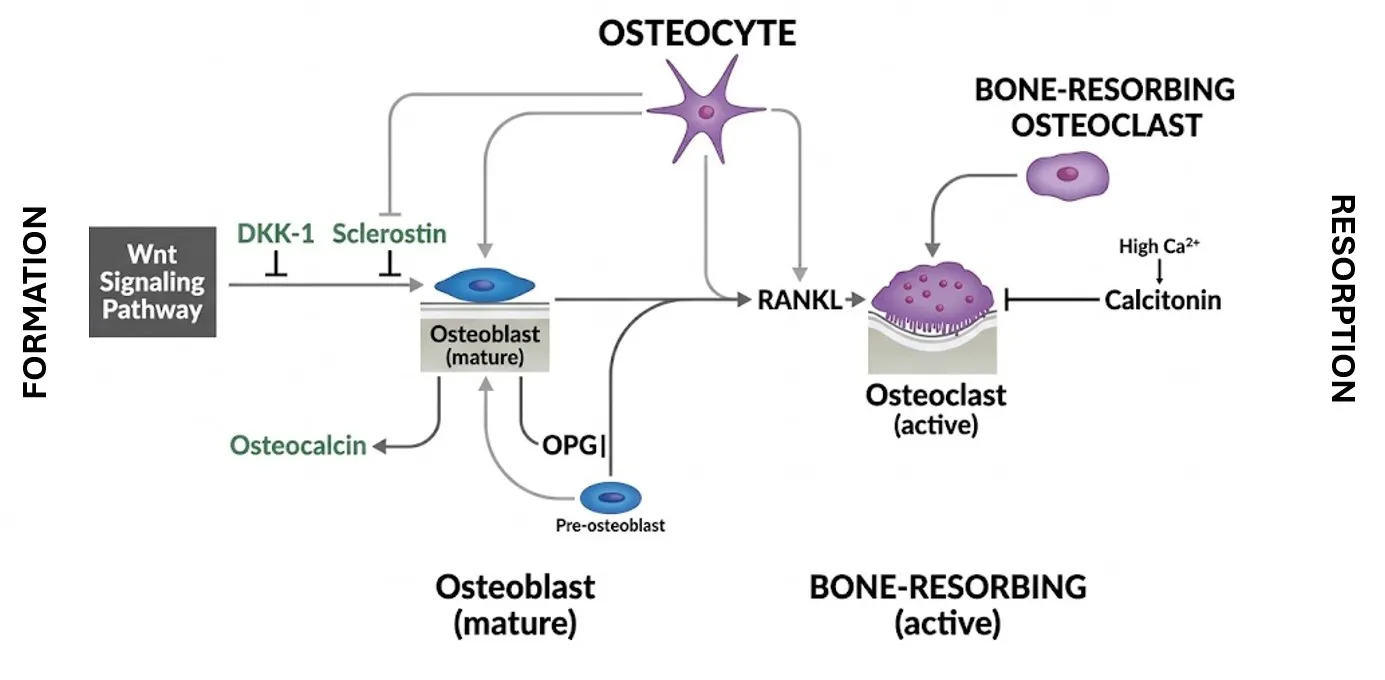
Figure 1: Biological pathway of bone tissue.
ALPCO
Bone density is one of the most integrated measurable outputs of remodeling biology and skeletal strength. Recent pooled analyses across randomized trials show that treatment-related increases in hip BMD are not just a correlation but confer a causal benefit; they correspond meaningfully with subsequent fracture reduction across multiple therapies.[6] For that reason, BMD has become a credible endpoint predictive of outcomes rather than just an exploratory biomarker. The main therapeutic strategies for osteoporosis focus on the two key components of the disease: excessive bone resorption and inadequate bone formation.
Primary Drug Class: Antiresorptive Treatment Options
The American College of Physicians (ACP) currently recommends antiresorptives (bisphosphonates and Anti-RANKL therapies) as the initial drug treatment for postmenopausal women and men with primary osteoporosis. Bisphosphonates work by slowing osteoclast activity and reducing bone resorption, and they remain the most prescribed treatment for osteoporosis. If bisphosphonates do not work for patients, the second recommendation is anti-RANKL therapies (e.g. denosumab), while sequential treatment (bone building anabolic agents followed by a bisphosphonate) is reserved for women at very high fracture risk.[7] This sequencing is clinically sensible, but it also highlights a central limitation of the field: the therapies most practical for widespread long-term use are not always the most effective at rebuilding bone.
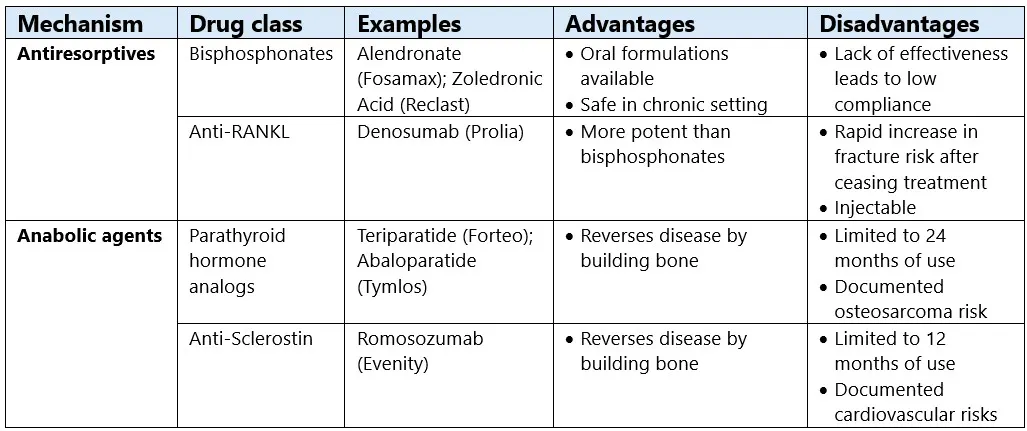
Bisphosphonates remain the backbone of care, as they reduce fractures, are familiar, and are cost-effective. But newer anabolic therapies outperform them in high-risk patients. In practice, antiresorptives are better for maintaining bone than rebuilding it, and persistent underuse of osteoporosis drugs reflects real-world concerns about their limitations and safety risks.
Anti-RANKL therapies illustrate both the strengths and the fragility of the antiresorptive strategy. It is effective and widely used, but its discontinuation profile is difficult to reconcile with a decades-long disease. The FDA label warns that after stopping Prolia, fracture risk increases, bone turnover rises above pretreatment values, BMD returns to pretreatment levels within 18 months, and new vertebral fractures have occurred as early as 7 months after the last dose. The 2024 label also added a boxed warning for severe hypocalcemia in patients with advanced kidney disease.[8] These issues do not mean denosumab lacks clinical value, but they do show why it is not a simple lifetime solution.
Primary Drug Class: Antiresorptive Treatment Options
In contrast to antiresorptives, anabolic agents, such as the parathyroid hormone analogs and anti-sclerostin therapies, target the other half of the disease: inadequate bone formation. Anabolic therapy reduces fractures faster, and to a greater extent, than antiresorptive therapy in high- and very-high-risk patients, but their concerning side effects and safety risks limit them to short-term use rather than long-term treatment.
The parathyroid hormone analogs show this tradeoff clearly. Teriparatide is administered as a once-daily subcutaneous injection, and its label states that use beyond two years in a patient’s lifetime should be considered only if the patient remains or again becomes high risk.[9] These agents can be clinically valuable, but a daily injectable therapy with lifetime duration limits is not an ideal backbone for population-scale management of a multidecade disease.
Additionally, the anti-sclerostin antibody romosozumab demonstrates both the promise and the limitations of the current anabolic wave. Its label warns of a potential increased risk of myocardial infarction, stroke, and cardiovascular death and limits use to 12 monthly doses because the anabolic effect wanes after that period. At the same time, the data show it can rebuild bone and reduce fractures, but the benefits fade once treatment stops. This highlights that while bone regrowth is possible, there still isn’t a long-term, durable treatment option.
Standard of Care in Osteoporosis Underserves the Patient Population
The standard of care in this field ultimately fails patients because it does not align with the disease time frame and clinical reality. Osteoporosis develops and persists over decades, but the most effective bone building therapies have lifetime caps that leave millions of patients at risk of fractures. Faced with this dilemma, clinicians and patients are stuck in a duality: antiresorptive therapies are ineffective and anabolic therapies are harder to use and cannot be used chronically—with no single therapy that has appropriate safety, efficacy, and durability.[10],[11] Balancing this duality works for high-risk patients as they are the only patient segment eligible for both antiresorptives and anabolic therapies, but this still leaves millions without feasible long-term treatment options. In large U.S. claims analyses, over 70% of patients with fragility fractures did not have documented osteoporosis.[12],[13] Even among post-menopausal women with confirmed osteoporosis, drug refill rates remain persistently low.[14],[15] Diagnosis and treatment in this area are still far from fully solved, with only marginal improvements achievable.
At a deeper level, current care still tends to manage decline rather than truly fixing the underlying problem (e.g. the remodeling balance). Antiresorptives are effective at slowing loss and preserving prior gains, but they do not fully address the loss of bone architecture accumulated over years. Bone building anabolic agents are effective at reversing the disease course, but the current anabolic therapies do not offer a durable, low-friction product that can be used beyond 1-2 years.[16],[17] The result is a treatment paradigm in which the field has meaningful drugs, but not a treatment regimen that combines the potency of the best anabolic agents with the safety, practicality, and durability required for widespread chronic use. Without that optimal product profile from a therapy, millions of patients continue to experience progressive bone weakening, increasing their fracture risk.
Regulatory Reset: From Development Friction to Practical Path Forward
There are 10s of millions of patients under addressed in the U.S. alone, representing a large market opportunity like those seen with the diabetes and weight loss drugs such as GLP-1s. Why haven’t biotech, pharma, scientists, and investors been developing drugs for these patients? The challenge in osteoporosis goes beyond the typical challenges of drug development: demanding regulatory requirements have stifled innovation.
For innovators, the second major obstacle beyond biology was the evidentiary burden. Under the FDA’s pre-2025 framework, BMD could support fracture-risk assessment, but it was not sufficient on its own to establish efficacy. Sponsors still had to demonstrate increases in BMD and reductions in fracture incidence in clinical trials. In practice, even when Phase 2 studies showed meaningful gains in BMD and other signs of skeletal improvement, those results were not registration-enabling. Companies still had to run very large, lengthy, and costly Phase 3 fracture-endpoint trials to prove that patients receiving treatment experienced a reduced disease burden.
The time, cost, and scale of these trials are rate-limiting. Even large pharmaceutical companies with the balance sheets to run these trials are too risk-averse to undertake them, and many concluded that the space was too challenging to justify continued investment in new drug programs. This dynamic was even more pronounced for smaller, more innovative biotechnology companies, which typically cannot (and in most cases should not) run trials of that magnitude. The burden of these studies stifled innovation by preventing promising programs from advancing.
However, it took more than a decade of persistent work by researchers and clinicians to influence regulators and reduce barriers to innovation. In 2020, a pooled individual-patient meta-regression of 91,779 participants across 23 randomized placebo-controlled trials found that treatment-related changes in BMD were significantly associated with reductions in vertebral, hip, and nonvertebral fractures. In that analysis, changes in hip BMD explained about 44% to 67% of fracture risk reduction, depending on fracture site.[18] A 2024 FNIH-ASBMR-SABRE analysis, using 122,235 participants from 22 randomized placebo-controlled trials, reinforced those findings, showing that treatment-related changes in total hip BMD at 12, 18, and 24 months were all associated with fracture risk reduction across trials, with stronger associations at longer intervals.[19] Together, these large cross-trial datasets helped move BMD from a plausible marker to a validated bridge between drug effect and clinical outcome.
The FDA translated that body of evidence into policy on December 19, 2025, qualifying percentage change from baseline at 24 months in total hip BMD measured by DXA as a validated surrogate endpoint for investigational therapies in postmenopausal women with osteoporosis at risk for fracture.[20] In the previous framework, Phase 2 and Phase 3 trials used different efficacy endpoints (BMD vs. fracture incidence). Moving forward, Phase 2 and Phase 3 trials will both use BMD as the primary efficacy endpoint. Early clinical trials will now generate more interpretable human efficacy signals with less capital, while companies have a clearer path from early signal to registrational strategy. We believe this to be a watershed moment in the development and treatment of osteoporosis that will make the space much more tractable for big pharma, biotech, and investors, spurring innovation over the next decade.

Osteoporosis at an Inflection Point: Improved Development, Unmet Biology
Osteoporosis is a large, chronic bone remodeling disease that has long been mismatched with both its therapeutic toolkit and its drug development framework. The field severely lacks a therapy that combines the potency of the best anabolic agents with the safety, practicality, and durability required for years or decades of treatment. That gap leaves millions of patients searching for answers, as patients’ bones continue to weaken despite long-term treatment. With current treatment options, it is not a matter of “if” a fracture will occur, but “when.” The previous innovation cycle has failed to meet the needs of patients, and a new era of drug development is on the horizon.
We are encouraged by the recent FDA changes that will usher in a new era of bone health-focused drugs. The SABRE project, which provided the scientific bridge, combined with the FDA’s 2025 qualification of total hip BMD as a validated surrogate endpoint for phase 3 trials, has made osteoporosis a more tractable category to develop than it was under the fracture-endpoint model.[22],[23] Although we have a long way to go on better uncovering the biological underpinnings of bone health, the improved regulatory environment is making osteoporosis a more likely area for innovation, and one in which we are actively seeking new science, teams, and companies.
- [1]Wright NC, Looker AC, Saag KG, et al. The recent prevalence of osteoporosis and low bone mass in the United States based on bone mineral density at the femoral neck or lumbar spine. J Bone Miner Res. 2014;29(11):2520-2526. doi:10.1002/jbmr.2269
- [2]U.S. Food and Drug Administration. FDA Qualifies Total Hip Bone Mineral Density (BMD) as Surrogate Endpoint for Osteoporosis Drug Development. FDA.gov. Published December 19, 2025.
- [4]Khosla S, Hofbauer LC. Osteoporosis treatment: recent developments and ongoing challenges. Lancet Diabetes Endocrinol. 2017;5(11):898–907.
- [5]Farr JN, Khosla S. Skeletal changes through the lifespan—from growth to senescence. Nat Rev Endocrinol. 2015;11(9):513-521.
- [7]Qaseem A, Hicks LA, Etxeandia-Ikobaltzeta I, et al. Pharmacologic Treatment of Primary Osteoporosis or Low Bone Mass to Prevent Fractures in Adults: A Living Clinical Guideline From the American College of Physicians. Ann Intern Med. 2023;176(2):224–238.
- [8]U.S. Food and Drug Administration. Prolia (denosumab) injection, for subcutaneous use [Prescribing Information]. Amgen Inc; 2024.
- [9]U.S. Food and Drug Administration. Forteo (teriparatide injection) [Prescribing Information]. Eli Lilly and Company; 2021.
- [10]Qaseem A, Hicks LA, Etxeandia-Ikobaltzeta I, et al. Pharmacologic Treatment of Primary Osteoporosis or Low Bone Mass to Prevent Fractures in Adults: A Living Clinical Guideline From the American College of Physicians. Ann Intern Med. 2023;176(2):224–238.
- [11]Wright NC, et al. The recent osteoporosis crisis in the United States: A review of the evidence, the causes, and our response. J Bone Miner Res. 2021;36(10):1889-1896.
- [12]Wright NC, et al. The recent osteoporosis crisis in the United States: A review of the evidence, the causes, and our response. J Bone Miner Res. 2021;36(10):1889-1896.
- [13]Gillespie CW, Morin PE. Trends and Disparities in Osteoporosis Screening Among Women in the United States, 2008-2014. Am J Med. 2017;130(3):306-316.
- [14]Wright NC, et al. The recent osteoporosis crisis in the United States: A review of the evidence, the causes, and our response. J Bone Miner Res. 2021;36(10):1889-1896.
- [15]Gillespie CW, Morin PE. Trends and Disparities in Osteoporosis Screening Among Women in the United States, 2008-2014. Am J Med. 2017;130(3):306-316.
- [16]Khosla S, Hofbauer LC. Osteoporosis treatment: recent developments and ongoing challenges. Lancet Diabetes Endocrinol. 2017;5(11):898–907.
- [17]Wright NC, et al. The recent osteoporosis crisis in the United States: A review of the evidence, the causes, and our response. J Bone Miner Res. 2021;36(10):1889-1896.
- [18]Black DM, Bauer DC, Vittinghoff E, et al. Treatment-related changes in bone mineral density as a surrogate biomarker for fracture risk reduction in clinical trials of osteoporosis treatment: A meta-regression analysis sponsored by the FNIH Bone Quality Project. J Bone Miner Res. 2020;35(12):2336-2348.
- [19]Vilaça T, Lui LY, Schini M, et al. Treatment-related changes in total hip bone mineral density are applicable to trials of varied study designs and to drugs with differing mechanisms of action: meta-regression results from the FNIH-ASBMR SABRE study. J Bone Miner Res. 2024;39(11):1228–1238.
- [20]U.S. Food and Drug Administration. FDA Qualifies Total Hip Bone Mineral Density (BMD) as Surrogate Endpoint for Osteoporosis Drug Development. FDA.gov. Published December 19, 2025.
- [21]Thompson A, Carroll K, Inrig JK, et al. Proteinuria Reduction as a Surrogate End Point in Trials of IgA Nephropathy. Clin J Am Soc Nephrol. 2019;14(3):469–481
- [22]U.S. Food and Drug Administration. FDA Qualifies Total Hip Bone Mineral Density (BMD) as Surrogate Endpoint for Osteoporosis Drug Development. FDA.gov. Published December 19, 2025.
- [23]Vilaça T, Lui LY, Schini M, et al. Treatment-related changes in total hip bone mineral density are applicable to trials of varied study designs and to drugs with differing mechanisms of action: meta-regression results from the FNIH-ASBMR SABRE study. J Bone Miner Res. 2024;39(11):1228–1238.