CEO Q&A: Curve Biosciences on Transforming Disease Detection Through Blood-Based Insights
By Luma Group - 5 minutes

At Luma Group, we partner with companies that are redefining the frontiers of healthcare through data, AI, and deep biological insight. Curve Biosciences is one such company, building a platform aimed at transforming how chronic diseases are detected and monitored. By turning a simple blood draw into a dynamic, organ-level view of disease biology, Curve is working to enable earlier intervention and more precise clinical decision-making.
We spoke with the CEO of Curve Biosciences, Dr. Ritish Patnaik, to discuss the company’s Whole-Body Intelligence platform, recent clinical progress in cirrhosis, and how advances in AI and genomic modeling are shaping the future of disease monitoring.
Q: For those who may not be familiar, can you give a quick overview of Curve Biosciences — what problem are you solving?
A: At Curve Biosciences, we’re addressing a fundamental limitation in modern medicine for chronic disease patients, the lack of non-invasive, serial visibility into what’s happening across the body at the organ level. Today, doctors rely on a combination of inaccessible imaging, outdated lab markers, and invasive biopsies, which are often episodic, indirect, and sometimes too late to capture early progression of chronic disease.
We aim to fundamentally shift that paradigm. We’re building a platform that transforms a simple blood draw into a dynamic, body-wide readout of both organ health and chronic disease stage through a deep understanding of biology across all tissues of the body — what we call “Whole-Body Intelligence.” By identifying early progression through more precise monitoring of chronic disease, we believe that we can help physicians intervene sooner, track chronic disease progression accurately, and improve patient outcomes.
Q: At a high level, how does your Whole-Body Intelligence platform work, and how are you able to read organ-specific signals from a simple blood test?
A: The body is constantly releasing molecular signals into the bloodstream—particularly cell-free DNA and RNA—from different tissues and organs. The challenge has been that these signals are highly complex and mixed together, making them difficult to interpret with traditional methods. For patients with chronic disease, this challenge is compounded by additional biological material coming from the target diseased organ and other distressed organs, which acts as confounding background noise in blood.
Our platform combines high-resolution molecular profiling with AI models (Whole-Body Intelligence models) trained on our proprietary Whole-Body Atlas covering over 400,000 samples across all the tissue types of the body. This atlas maps DNA in the blood back to their tissue and cellular origins using their methylation patterns. With that framework, we can deconvolute blood samples and identify which organs are contributing specific signals, and what those signals indicate about biological activity, stress, or disease.
In practical terms, that means a single blood test can provide organ-specific insights with greater precision —whether that’s the liver, kidney, or other organs—without the need for invasive procedures.
Q: Let’s start with the cirrhosis study—without getting ahead of the full publication later this year. What can you share about the key takeaways so far?
A: What we’re seeing so far is very encouraging. The platform is able to detect biologically meaningful changes associated with disease progression in liver cirrhosis patients, and those signals correlate strongly with clinical outcomes.
One of the most important observations is that we’re capturing changes earlier and more dynamically than many traditional monitoring approaches. Cirrhosis is a disease where patients can remain relatively stable and then deteriorate quickly, and existing tools often don’t provide sufficient lead time. Our data suggest we may be able to identify subtle shifts in disease trajectory before they become clinically apparent.
Q: You evaluated 1,482 patients across 22 sites — how does that scale strengthen confidence in the results?
A: Scale is critical for both scientific rigor and clinical relevance. By including nearly 1,500 patients across 22 sites, we capture a wide range of real-world variability — differences in patient demographics, disease severity, treatment patterns, and care settings.
That diversity helps ensure that the signals we’re detecting are not artifacts of a single institution or a narrowly defined cohort. Instead, it gives us confidence that the platform is identifying robust, reproducible biological patterns that can generalize across broader patient populations.
For this clinical need, sensitivity, or accuracy of the detection of progression, is the most valued performance metric. To our knowledge, our completed study is the largest ever performed in terms of the number of cirrhosis patients who progressed to liver cancer compared to other blood-based liver cirrhosis monitoring tests. This size advantage also allows us to understand the sensitivity advantage of our blood test with high confidence.
Q: At a high level, how did your approach perform relative to current standard-of-care monitoring?
A: While we’ll present full results in the upcoming publication, at a high level, our approach demonstrated the potential to outperform the standard-of-care method, as well as other tests in development, in detecting and tracking disease progression.
Traditional tools often rely on indirect or lagging indicators of liver function and anatomy. In contrast, our platform is measuring underlying biological activity more directly, which allows for a more sensitive and real-time view of what’s happening in the liver. That difference is what we believe drives the improved performance we’re seeing.
Q: You’ve talked about filtering biological noise—how important was that in achieving the performance you’re seeing?
A: It’s absolutely essential. Blood is an incredibly information-rich but noisy environment. Signals from multiple organs are mixed together, and there’s a significant amount of background variability between individuals.
A key part of our Whole-Body Intelligence approach is leveraging AI to separate the true biological signals from noise. By training our AI models on large, well-curated datasets, we identify consistent patterns that reflect real organ-specific biology, while filtering out irrelevant variation.
Without that layer of signal refinement, it is very difficult to achieve the level of sensitivity and specificity that we’re seeing, as we’ve seen in studies for other cirrhosis blood tests.
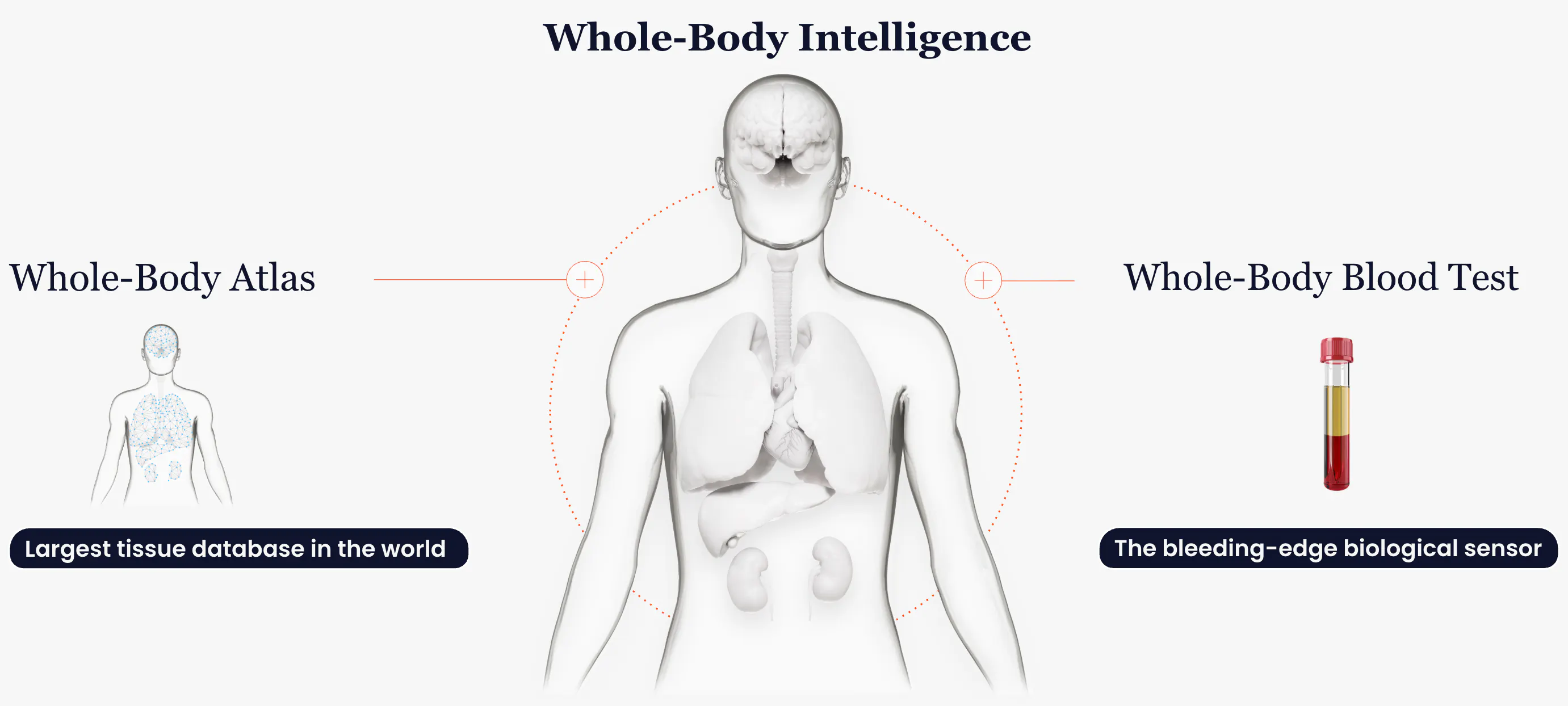
Curve Biosciences' Whole Body Intelligence Diagram
Curve Biosciences.
Q: From a clinical standpoint, how could this change how physicians monitor cirrhosis patients once fully validated?
A: We believe that our test will represent a shift from episodic, reactive monitoring to more continuous and proactive cirrhosis management.
Instead of waiting for clinical deterioration or relying on intermittent, inconsistent lab test values, hepatologists can use a blood-based test to track disease biology over time and identify early progression. That can inform more timely interventions, closer monitoring, and personalized treatment. Proving this in cirrhosis would add momentum to bringing these clinical improvements to other chronic disease patients and their physicians.
Q: What does earlier detection of disease progression enable in terms of intervention or patient outcomes?
A: Earlier chronic disease progression detection creates a wider window of opportunity for patients. In cirrhosis, once patients decompensate or develop large tumors, outcomes worsen significantly and treatment options become more limited.
If you can identify liver disease progression earlier, you can intervene sooner—whether that’s adjusting therapy, prioritizing patients for liver transplant evaluation, or increasing surveillance to prevent complications. Ultimately, the goal is to shift the disease course before irreversible damage occurs.
Q: How are you thinking about expanding into related areas like MASH (fatty liver inflammation) or broader liver diseases?
A: Cirrhosis is just one part of a much broader continuum of liver disease. We see a significant opportunity to apply the same platform to earlier stages, including MASH, where there is a major unmet need for non-invasive diagnostics and monitoring tools.
Because our approach is fundamentally based on reading organ-specific signals, it’s inherently extensible. We’re actively exploring how to expand across the liver disease spectrum and, more broadly, into other organ systems where similar challenges exist.
Q: On the AI side, some of your genomic foundation model work was accepted to ICLR—what was the core innovation behind that work?
A: We disclosed some methods that allow us to retrofit genomic language models to DNA methylation foundation models via continual pretraining. Beyond feasibility, we looked at interpretability of resulting models, as well as how informative and expensive they were.
We found that this strategy led to informative methylation-aware foundation models that were interpretable and inexpensive if high-quality input DNA methylation data was used. These models can also be adapted across multiple use cases—from disease detection to tissue-of-origin mapping—while benefiting from shared learning from large datasets.
Q: How tightly integrated are your foundation models with the Whole-Body Atlas—does performance improve as the dataset grows?
A: They’re becoming more and more deeply integrated. The Whole-Body Atlas provides the structured, high-quality data that the foundation models are trained on, and in turn, the models can help refine and expand the atlas.
As the dataset grows in both scale and diversity, we will see continuous improvements in model performance. More high-quality data allows models to capture more nuanced biological patterns, improve accuracy, and extend our capabilities to new indications.
Q: What have you learned about the role of high-quality, curated data in building clinically useful AI models?
A: It’s the most important factor for us. An AI model’s performance is only as good as the input data it’s trained on. High-quality, well-annotated, and clinically relevant datasets are critical for building models in healthcare that perform reliably in real-world settings.
We’ve invested heavily in data curation and quality control with our Whole-Body Atlas, because that’s what ultimately enables us to generate insights that clinicians can trust.
Q: You recently brought on Amit Singal as Chief Medical Officer—what drove that decision at this stage?
A: As we transition from platform validation to large-scale clinical studies and publication, it was critical to bring in a leader with deep expertise and credibility in liver disease. Amit is widely recognized as one of the leading authorities in hepatology, with a career that has focused on improving outcomes for patients with MASH, cirrhosis, and liver cancer.
He currently serves as Chief of Hepatology at UT Southwestern, where he leads both clinical care and research efforts across the liver disease continuum—from prevention and screening to early detection and treatment. He also serves as Medical Director of the UT Southwestern Liver Tumor Program and holds the Willis C. Maddrey Distinguished Chair in Liver Disease.
His work has been particularly focused on closing gaps in care for cirrhosis patients, including identifying disease progression and liver cancer earlier, when intervention is still possible. He has authored more than 300 peer-reviewed publications and led multiple NIH-funded studies examining the cirrhosis-cancer care continuum.
That combination of frontline clinical experience, translational research leadership, and a deep understanding of real-world care pathways made him the ideal partner for us as we move further towards clinical validation and adoption.
Q: What role will he play as you move toward publication, validation, and eventual clinical adoption?
A: Amit plays a central role in shaping our clinical strategy and ensuring that what we’re building aligns with how medicine is actually practiced. He is leading efforts around study design, endpoint selection, and interpretation of clinical data as we prepare for publication and broader validation.
Equally important, he brings a strong perspective on how new tools need to integrate into clinical workflows to truly impact patient care. His experience leading large liver disease programs and managing complex patient populations gives him a clear view on where current approaches fall short—and where we can make the biggest difference.
He is also deeply connected to the hepatology community, which is key as we engage with clinicians, build trust in the data, and ultimately drive adoption. At a broader level, his focus aligns closely with our mission: shifting liver disease progression detection and management earlier, when there is still opportunity to meaningfully change outcomes.
Q: With full results expected later this year, what milestones should we be watching for?
A: The critical milestone will be the full publication of our cirrhosis study. We also plan to conduct a final cirrhosis study, expand to additional liver and non-liver indications, and further advance our AI models and Whole-Body Atlas. Each of these steps is focused on moving Curve closer to clinical adoption and real-world impact.